Health
Enzyme Mutation Uncovers New Insights into Dementia Mechanism
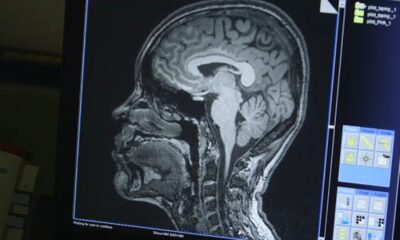
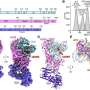
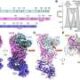
Researchers at Helmholtz Munich have identified a crucial enzyme mutation that acts as a hidden trigger for early-onset dementia. This discovery, published on December 9, 2025, in the journal *Cell*, reveals how the enzyme *glutathione peroxidase 4* (GPX4) protects neurons, and how a rare mutation disrupts this protective mechanism, leading to neuronal damage similar to that seen in Alzheimer’s disease.
The study, led by Prof. Marcus Conrad, Director of the Institute of Metabolism and Cell Death at Helmholtz Munich, centers on the structural features of GPX4. Researchers found that a specific genetic change in this enzyme can remove its ability to shield neurons from damage caused by lipid peroxides, harmful molecules that can compromise cell membranes. The mutation, known as R152H, has been identified in children suffering from a severe form of early-onset dementia.
Understanding the protective role of GPX4 is critical. When functioning correctly, GPX4 features a small protein loop, likened to a “fin,” that inserts itself into the inner surface of neuronal membranes. This positioning enables the enzyme to efficiently detoxify lipid peroxides. As Prof. Conrad explains, “GPX4 is a bit like a surfboard. With its fin immersed into the cell membrane, it rides along the inner surface and swiftly detoxifies lipid peroxides as it goes.”
In children with the R152H mutation, the fin-like loop is altered, preventing GPX4 from embedding correctly in the membrane. This impairment allows lipid peroxides to accumulate, leading to a process called ferroptosis, which results in cell rupture and neuronal loss.
The research began with three children from the United States, all of whom share the R152H mutation. Scientists derived cells from one child and reverted them to a stem-cell-like state. These cells were then used to grow cortical neurons and three-dimensional brain organoids, providing a platform to study the mutation’s effects.
To investigate the mutation’s impact on a whole organism, researchers introduced the R152H variant into a mouse model. Mice with this mutation exhibited significant motor problems, neuron loss in key brain regions, and strong neuroinflammatory responses. These findings closely mirrored the symptoms observed in the affected children and exhibited patterns similar to those found in other neurodegenerative diseases.
Analysis of protein levels in the experimental model revealed shifts analogous to those documented in Alzheimer’s disease. Many proteins that show altered levels in Alzheimer’s patients also exhibited similar disruptions in the mice lacking functional GPX4. This connection suggests that ferroptotic stress could play a role not only in this rare childhood condition but also in more prevalent forms of dementia.
Dr. Svenja Lorenz, one of the study’s first authors, noted, “Our data indicate that ferroptosis can be a driving force behind neuronal death — not just a side effect.” This perspective shifts the focus of dementia research, which has traditionally concentrated on amyloid beta plaques, to the damage occurring at the cellular membrane level that initiates neurodegeneration.
Initial tests indicate that targeting ferroptosis can slow cell death associated with the loss of GPX4, both in cell cultures and in the mouse model. Dr. Tobias Seibt, a nephrologist at LMU University Hospital Munich and co-first author, emphasizes that while this is a significant proof of principle, it is not yet a therapeutic solution.
Dr. Adam Wahida, another first author of the study, suggested that future research could explore genetic or molecular strategies to stabilize the protective mechanisms of GPX4. However, he acknowledged that the current findings primarily contribute to basic research.
The project represents a culmination of nearly 14 years of collaborative scientific effort, involving expertise from genetics, structural biology, stem cell research, and neuroscience. Prof. Conrad stated, “Projects like this vividly demonstrate why we need long-term funding for basic research and international multidisciplinary teams if we are to truly understand complex diseases such as dementia and other neurodegenerative conditions.”
This groundbreaking research not only enhances understanding of dementia mechanisms but also opens new avenues for therapeutic exploration, potentially paving the way for innovative treatments in the future.
-

 Science4 months ago
Science4 months agoNostradamus’ 2026 Predictions: Star Death and Dark Events Loom
-

 Science4 months ago
Science4 months agoBreakthroughs and Challenges Await Science in 2026
-

 Technology8 months ago
Technology8 months agoElectric Moto Influencer Surronster Arrested in Tijuana
-

 Technology5 months ago
Technology5 months agoOpenAI to Implement Age Verification for ChatGPT by December 2025
-

 Technology10 months ago
Technology10 months agoDiscover the Top 10 Calorie Counting Apps of 2025
-

 Health8 months ago
Health8 months agoBella Hadid Shares Health Update After Treatment for Lyme Disease
-

 Health8 months ago
Health8 months agoAnalysts Project Stronger Growth for Apple’s iPhone 17 Lineup
-

 Health8 months ago
Health8 months agoJapanese Study Finds Rose Oil Can Increase Brain Gray Matter
-

 Technology4 months ago
Technology4 months agoTop 10 Penny Stocks to Watch in 2026 for Strong Returns
-

 Science7 months ago
Science7 months agoStarship V3 Set for 2026 Launch After Successful Final Test of Version 2
-

 Technology7 months ago
Technology7 months agoInMotion Unveils P6 Electric Unicycle with 93 MPH Top Speed
-

 Technology2 months ago
Technology2 months agoNvidia GTC 2026: Major Announcements Expected for AI and Hardware