Health
BeOne Advances Rare Lymphoma Drug After Promising Trial Results
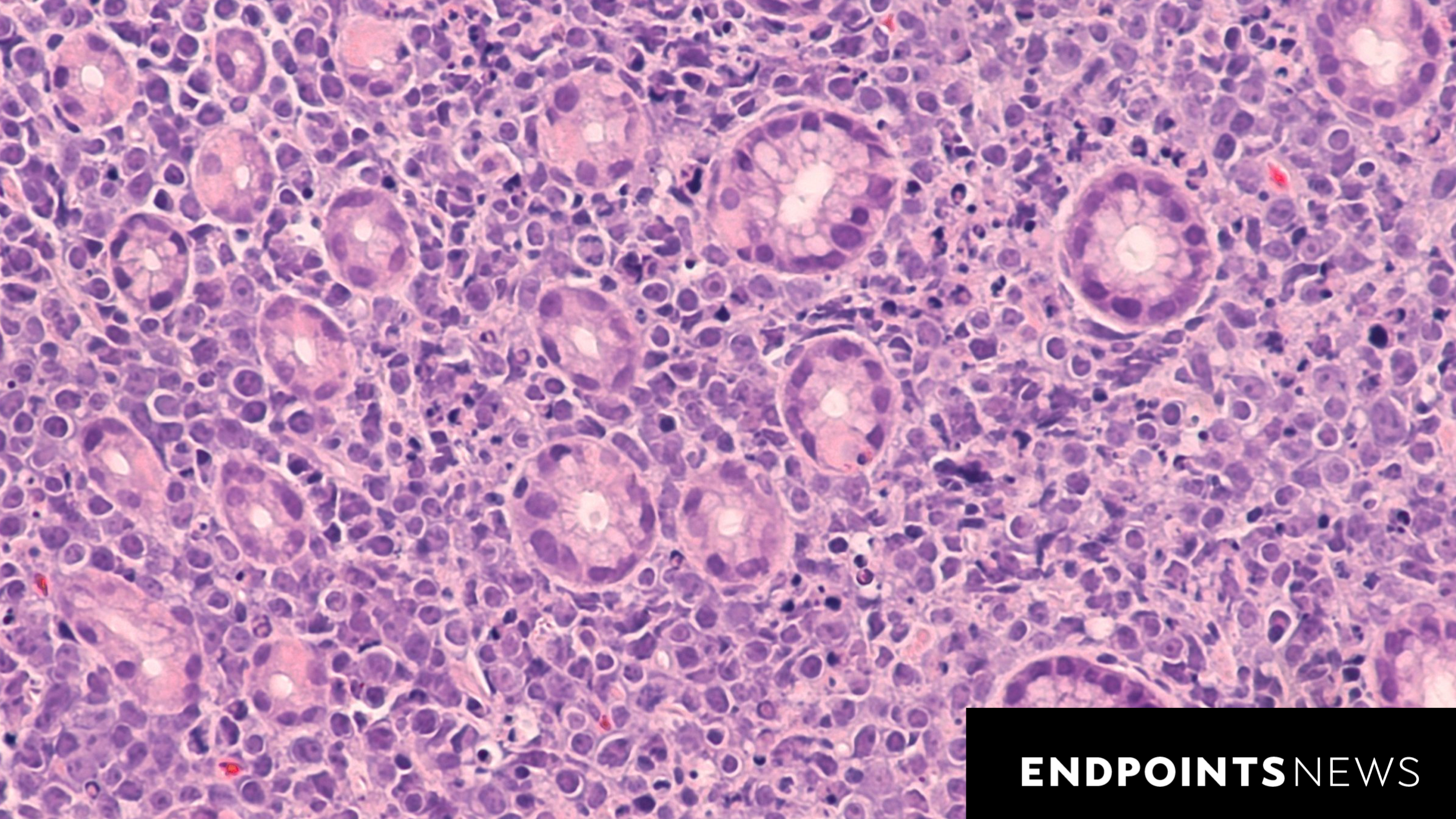
BeOne Medicines has announced significant progress in the development of its drug candidate for a rare and aggressive form of B cell lymphoma. The company reported that its BCL2 inhibitor successfully passed early-stage clinical trials, paving the way for potential accelerated approval from regulatory authorities.
The Phase 1/2 clinical trial demonstrated promising results, indicating the drug’s ability to target specific cancer cells effectively. This outcome positions BeOne to seek expedited approval from the U.S. Food and Drug Administration (FDA), which could significantly shorten the timeline for bringing this treatment to patients in need.
Details of the Clinical Trial
The clinical trial involved a diverse group of participants diagnosed with aggressive B cell lymphoma. The primary focus was to assess the safety and efficacy of the BCL2 inhibitor. According to BeOne, the results showed a favorable safety profile, with many participants experiencing a reduction in tumor size after treatment.
BeOne’s Chief Executive Officer, Dr. Sarah Thompson, expressed enthusiasm about the results, stating, “The positive outcomes from our early-stage trial reaffirm our commitment to addressing unmet medical needs in oncology.”
The company plans to submit its findings to the FDA by March 2024, aiming for a swift review process. If the FDA grants accelerated approval, it would mark a significant milestone for BeOne and the patients affected by this challenging form of cancer.
Market Impact and Future Prospects
The potential approval of BeOne’s drug could not only benefit patients but also create substantial market opportunities. The global market for lymphoma treatments is expected to grow significantly, driven by increasing incidence rates and the demand for innovative therapies. Analysts project that the BCL2 inhibitor could capture a notable share of this market, particularly if it proves effective in larger clinical trials.
Investors are closely monitoring BeOne’s progress as the company navigates the regulatory landscape. The successful completion of the Phase 1/2 trial has already sparked interest, with shares of BeOne showing an uptick following the announcement.
As BeOne prepares for the next steps, stakeholders in the healthcare sector will be watching closely. The outcome of the FDA review could set a precedent for future oncology treatments and highlight the importance of rapid innovation in the pharmaceutical industry.
The journey from trial results to market availability is often complex, but BeOne Medicines is positioned to lead the charge in addressing the challenges of rare lymphomas. The commitment to advancing patient care remains at the forefront of their mission as they look toward the future.
-

 Technology4 months ago
Technology4 months agoDiscover the Top 10 Calorie Counting Apps of 2025
-

 Health2 months ago
Health2 months agoBella Hadid Shares Health Update After Treatment for Lyme Disease
-

 Health3 months ago
Health3 months agoErin Bates Shares Recovery Update Following Sepsis Complications
-

 Technology3 weeks ago
Technology3 weeks agoDiscover 2025’s Top GPUs for Exceptional 4K Gaming Performance
-

 Technology2 months ago
Technology2 months agoElectric Moto Influencer Surronster Arrested in Tijuana
-

 Technology4 months ago
Technology4 months agoDiscover How to Reverse Image Search Using ChatGPT Effortlessly
-

 Technology4 months ago
Technology4 months agoMeta Initiates $60B AI Data Center Expansion, Starting in Ohio
-
 Technology4 months ago
Technology4 months agoRecovering a Suspended TikTok Account: A Step-by-Step Guide
-

 Health4 months ago
Health4 months agoTested: Rab Firewall Mountain Jacket Survives Harsh Conditions
-

 Lifestyle4 months ago
Lifestyle4 months agoBelton Family Reunites After Daughter Survives Hill Country Floods
-

 Technology3 months ago
Technology3 months agoUncovering the Top Five Most Challenging Motorcycles to Ride
-

 Technology4 weeks ago
Technology4 weeks agoDiscover the Best Wireless Earbuds for Every Lifestyle